
Quality System Implementation and Management
New Product Introduction
Product Development
Prospect New Product Introduction (NPI) is your trusted partner in bringing innovative medical devices to market. We specialize in the production of sterile instruments, diagnostic kits, combination devices, and electromechanical devices for the medical device and biotech industries. Our strong quality system, audited to ISO 13485:2016 and 21 CFR 820 (FDA QSR) regulations, ensures that your products meet the highest standards of compliance and reliability. With FDA registration (3014680149) as a contract medical device developer and manufacturer, we cover all aspects of medical device development, manufacturing transfer, and production, providing you with a seamless and efficient path to market success.
Device Design and Deliverables
We offer comprehensive device design services that turn medical concepts into manufacturable products. Our process covers everything from risk assessment to detailed design, prototyping, and design transfer, ensuring alignment with user needs, regulatory standards, and performance goals. Complementing this, our design deliverables include complete CAD models, engineering drawings, and verified specifications.
Design History File (DHF) and Technical Document Development and Remediation
We excel in developing and remediating Design History Files (DHFs) and technical documents to ensure your medical devices meet the highest standards of quality and compliance. Our meticulous approach guarantees that every essential document is in place, providing a solid foundation for regulatory approval and market success.
Risk Management & Regulatory Clearance
We implement a comprehensive risk management process consistent with ISO 14971, integrating hazard identification, risk analysis, and control measures throughout the development cycle. Our team prepares and maintains detailed risk files, design history documentation, and regulatory submission packages to support 510(k), PMA, or CE marking pathways, ensuring full compliance with global regulatory requirements.
Usability & Design Verification & Validation
We apply structured human factors and engineering methodologies to verify that the device design meets specified requirements and intended use. Our verification and validation activities include protocol development, test execution, data analysis, and reporting in accordance with FDA and ISO 13485 standards—confirming design performance, user safety, and readiness for design transfer.
Quality System Implementation and Management
We implement and manage robust quality systems that ensures every process is streamlined, documented, and compliant. Our comprehensive approach guarantees that every aspect of your quality system is optimized for efficiency and effectiveness, providing a solid foundation for your product’s success in the market.
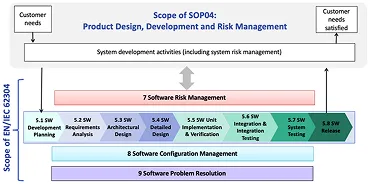
Software Development Life Cycle
We manage the full Software Development Life Cycle (SDLC) with diligence—from requirements engineering and risk management
through architecture, development, verification, validation, release, and post-market maintenance. Our workflows are optimized for
Software in a Medical Device (SiMD), aligning with IEC 62304 and FDA guidance to ensure traceability, compliance, and robust
performance across all lifecycle stages.




Visit Us
Westminster, Colorado
Contact us



