Innovate. Produce. Impact.
Your Medical Device Journey,
Simplified.
From initial concept to global market fulfilment, Prospect Life Sciences provides seamless, integrated solutions for medical device development and manufacturing, empowering innovations that enhance lives.
Explore our capabilities



Your Seamless Journey, Expertly Guided
We've integrated over a decade of expertise in research, design, and manufacturing to offer a truly unified pathway for your medical device. We simplify complexity, guiding your innovation from idea to impact.

Get a no-cost roadmap to FDA Clearance, CE Marking, or Quality System Implementation.
Unlock a free roadmap to streamline your product's journey, from concept to clearance. Contact us for a detailed Project Proposal Budget & Schedule.
Get your free roadmapGet your Premium roadmap
Our End-to-End Medical Device Pathway
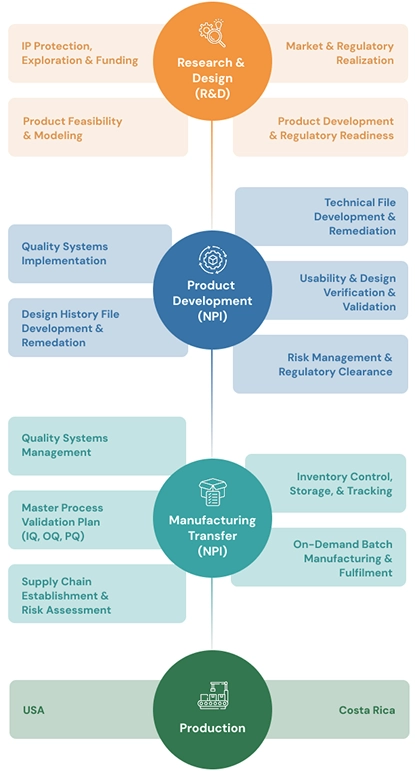
Integrated Success: From Idea to Impact.
We bridge the infamous "Valley of Death" between development and production, ensuring your medical device's seamless path to market, growth, and sustained impact.
Uncompromising Quality, Global Compliance.
Our ISO 13485:2016 certified and FDA registered Quality Management System is embedded at every stage, upholding the highest standards from design to final product.
Scalable Manufacturing, Strategic Reach.
Gain unparalleled flexibility with high-precision US manufacturing and cost-effective, high-volume production in Costa Rica, optimizing for efficiency and market access.

Accelerate Your Medical Device to Market.
Precision Manufacturing & Scaled Production, Where Innovation Meets Execution.
- Manufacturing in the USA & Costa Rica for strategic advantage.
- ISO 13485 Certified & FDA Registered Production.
- Full Lifecycle Support: From NPI to High-Volume Fulfillment.
Visit Us
11025 Dover Street
Westminster, Colorado
Westminster, Colorado
Contact us
We want to hear from you.



